Testosterone has a crucial, if poorly understood, effect on male behaviour. It contributes to aggressivenes, libido, tumescence and sexual performance. Some scientists believe that the ratio of index finger length to ring finger length indicates how much testosterone we were exposed to in our mother’s womb. This has led some Palm reader’s to use clues from the ‘index finger:ring finger’ ratio to deduce gendered behavioural characteristics of a client… hmmmm???
Concretely, what we do know is that Testosterone levels in early brain development, among many interesting things, can influence laterality, playing a role in handedness and the degree of linguistic lateralisation. In males, testosterone has many direct effects on the anatomy and metabolism. Male humans are characterised by strong bones, increased muscle mass and a deeper voice (although the aging elderly male voice actually rises in pitch). Testosterone stimulates the growth of the genitals at puberty and is responsible for sperm production throughout adult life. Testosterone, arguably also plays a role in male intelligence–(or lack thereof)! 😛
Testosterone might put hairs on your chest, but it can also contribute to male-pattern baldness and prostate disease. It is a funny little hormone that influences cholesterol metabolism, the production of red blood cells by bone marrow, secondary sex characteristics, musculature, weight, accessory organs, mortality and injury rates. It is sometimes over-popularised for what are actually poorly understood processes, but in recent research, testosterone may be an important factor in understanding plasticity in the brain!!! In this exciting discovery, researchers are beginning to understand a pivotal role testosterone is playing in neurochemical plasticity!
So, the time has come, (as the Walrus said to the Carpenter), to draw your attention to this recent publication which looks at testosterone with respect to environmental influences (the light-dark/sleep-wake cycle) and it’s effects within the brain of a seasonal mammal, the Djungarian hamster (Phodopus sungorus).
*SIGH* Ah, the beauty! A study of the brain in context!!!
While many researchers are looking at how to regenerate neurons (which could potentially help stroke victims, paraplegics and alzheimers patients etc), a small group of researchers at the Laboratoire de Neurobiologie des Rythmes, Universite Louis Pasteur, are looking at the role of testosterone in neurochemical plasticity. It is a significant step towards understanding how to guide freshly generated neurons! Regenerating neurons is only part of the journey for accident-recovery patients, guiding these neurons might be tricky and Testosterone may be an important key!
The context of the brain; The biology of a day.
« Scientists have broken down many kinds of systems…the next task is to reassemble them »
Edward O. Wilson, Professor Emeritus of Harvard University
Consilience: The Unity of knowledge, Random House, 1999.
The length of a day represents the time taken for the earth to complete one revolution about its vertical axis. The sun regulates the hours of daylight, and, from a biological standpoint, what we are concerned with is the time that it takes for the earth to rotate once with respect to the sun.
A year represents the time it takes for the earth to orbit the sun. For our biology, it is the timing of the seasons that is important. The seasons of the year are determined by the distance from the equator and the angle of tilt to the earth’s axis with respect to its plane of rotation about the sun.
Animals have developed many incredible physiological adaptations to cope with seasonal environmental extremes. Since reproductive fitness is the ultimate key to the survival of a species, studies of circannual rhythms have often focussed on the reproductive system, but this cannot function in isolation. It requires the support of other body systems, such as those concerned with hibernation, the ingestion and digestion of food, metabolism, thermoregulation, migration and behaviour
In mammals that respond to changes in the length of a day, there are a number of photoperiodically induced-responses:
- Reproductive state and associated behavioural changes
- Onset of and arousal from hibernation
- Body weight
- Thyroid or brown adipose tissue activities throughout the year
- Changes in fur colour, moulting
- Thermoregulatory capacities
- Migration
- Antler growth
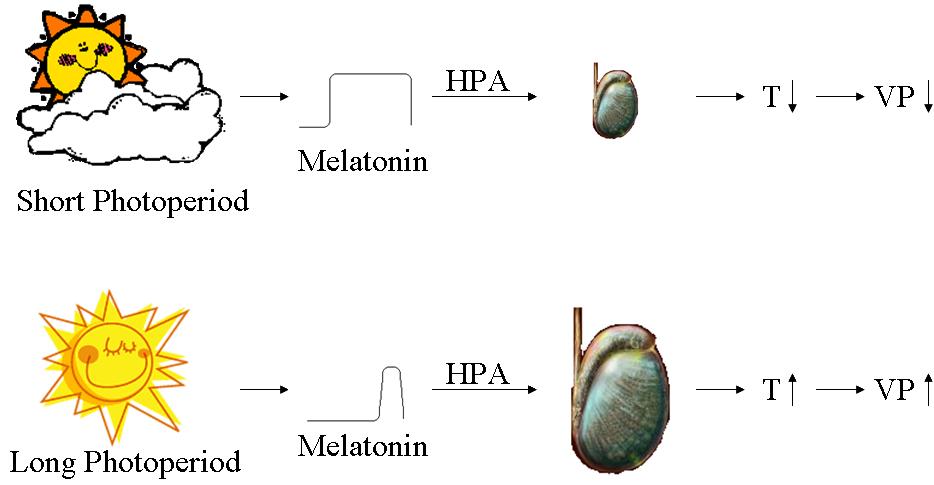
Animals use changes in the length of daylight, the photoperiod, to help predict the changing seasons. Melatonin via its rhythmic pattern of secretion is the, or one of, the pineal hormones responsible for transmitting daylength information. Daylight information enters the Supra-Chiasmatic Nucleus (SCN) from the retina and via the retino-hypothalamic tract and indirectly via the geniculo-hypothalamic tract originating in the Intergeniculate Leaflet. The SCN is known to play a crucial role via its connection to the pineal gland in the pathway mediating these photoperiod-induced reproductive changes.
The sex steroid hormones show photoperiodic changes in seasonal breeders. Thyroid hormones have also been shown to display photoperiodic variations. Hypothalamic Vasoactive intestinal peptide (VIP) and neuropeptide Y (NPY) synthesis is known to be influenced by thyroid hormones and in particular by thyroxin. This hormonal system may thus be considered as a factor in the sex steroid-induced modulation of NPY in the geniculohypothalamic tract as well as that of VIP within the SCN.
During long photoperiod seasonal breeders (like the Siberian hamster) are sexually active, whereas in the short photoperiod of autumn and winter, via an alteration of the daily pattern of melatonin secretion, acting on the hypothalamus and the pituitary, the gonads atrophy (see picture below), and the animals become sexually inactive. The subsequent reduction in testosterone levels induces changes in the central Vasopressin innervation of some brain areas.
HPA = Hypothalamic-Pituitary-Adrenal axis
T = Testosterone
VP = Vasopressin
During short photoperiod (winter) there is a dramatic reduction in the expression of vasopressin mRNA and protein in the Nucleus of the Stria Terminalis (NST). This results in reduced vasopressin immunoreactivity in the nerve fibres projecting from the NST to the lateral septum and indeed a marked disappearance of VP has been observed in the in the lateral septum of the European Hamster. At the end of winter, and possibly after several months of hibernation, the photoperiod starts to lengthen and the process is reversed. The gonads increase again in weight and circulating testosterone levels increase. As a consequence, there is also a progressive re-establishment of VP innervation. Similar seasonal variations in the expression of other neuropeptides can be observed in other systems.
To understand the brain mechanisms involved in the photoperiodic control of seasonal functions, the VP system should be considered first.
Vasopressin
Vasopressin (VP) influences a variety of sex-typical and species-specific behaviours and provides an integrational neural substrate for the dynamic modulation of those behaviours by endocrine and sensory stimuli. There are four major VP systems in the brain:
- 1. The autonomic vasopressin system, located in the PVN and involved in the control of several autonomic functions.
- 2. The classical neuroendocrine vasopressin system composed of the magnocellular neurons of the SON and the PVN.
- 3. a sexually dimorphic vasopressin system located in the NST and Amygdala. These neurons project among other areas to the lateral septum.The vasopressinergic projections of the principal nucleus of the bed nuclei of the stria terminalis are controlled by sex steroids. The VP projections are much more developed and larger in the male than in the female. The NST conveys olfactory information relevant for reproduction to the hypothalamus. There is a sexually dimorphic expression of neurohypophysial peptide receptors and a steroid dependency of vasopressin receptor expression.The sexual dimorphism in the ventromedial hypothalamic nucleus is affected by the length of the photoperiod and thus by gonadal steroids. Many neuropeptide systems show dramatic fluctuations in their expression under the influence of gonadal steroids; however, a complete elimination of the expression of particular neuropeptide by gonadectomy has been reported only for the VP cells of the BNST and Amygdala.
- 4. The biological clock vasopressin system, located in the dorso-medial part or the shell of the SCN.
AVP influences a variety of sex-typical and species-specific behaviours and provide an integrational neural substrate for the dynamic modulation of those behaviours by endocrine and sensory stimuli.
AVP is more extensively associated with male behaviour than with female behaviour, although this may partially reflect an emphasis on behaviours which are exhibited by males. Whilst AVP modulates a variety of behaviours exhibited only by males, oxytocin is extensively linked to female-specific functions.
Multiple vertebrate classes exhibit sex dimorphisms and steroid sensitivities for AVP neural elements in the septum, Bed Nucleus of the Stria Terminalis (BNST), periaqueductal gray, amygdala and pre-optic area anterior hypothalamus. These sites have also implicated in social behaviour. The behavioural actions of AVP may occur via a paracrine peptide action.
In rats and mice, it is the sexually dimorphic vasopressin system of the BNST that is the most sensitive neurotransmitter system regulated by sex steroids. In addition to vasopressin, the BNST neurons also express a second neuropeptide, galanin, whose expression also appears to be regulated by testosterone in laboratory rodents. Seasonal fluctuations of sex steroids in photoperiodic rodents feed back on the brain to regulate the expression of sex steroid sensitive genes. The seasonal rhythm of circulating sex steroids is generated by photoperiod-controlled melatonin secretion, resulting in a seasonal stimulation and involution of the gonads.
The seasonal changes in the expression of neuropeptides represents a neurochemical plasticity. The NST neurons project to the lateral septum, as do the Amygdala neurons. The reduction in vasopressin immunoreactivity in the lateral septum can be explained by the reduction in the synthesis and transport of vasopressin. However, in parallel to the reduction of vasopressin synthesis and transport, the nerve fibres are probably retracting at least partially from the lateral septum.
Seasonal structural plasticity is very well known in song birds, where several brain structures degenerate in winter, and re-establish a new functional system in spring with the generation of new neurons, migration of these neurons to different brain structures, and reconnection of these neurons into the neural network through axonal and dendritic growth and synaptogenesis.
In mammals, seasonal structural plasticity with axonal and dendritic growth followed by synaptogenesis has not been evaluated prior to this current research. Thus, seasonal VP expression in the NST, Amygdala and LS provides us with an excellent opportunity to study this phenomenon in mammals.
« The genetic and exogenetic components of behaviour
are different but interacting parts of a single system.
Human behaviour cannot be understood in
the absence of any of those components. »
Derek Freeman, 1983
It is important to note that the four Vasopressin systems mentioned above are somewhat contrived categories, and necessarily so for research purposes, but they are in reality extensively overlapping. Furthermore, the actions of Vasopressin are several and varied across species:
Actions of VP:
- Learning and memory functions
- Hibernation
- Grooming behaviour
- Exploratory/anxiety related behavior in rodents
- Sleep site investigation (fish)
- Locomotion (amphibians)
Vasopressin also modulates a range of social behaviours:
- Olfactory (scent marking) and vocal communication
- Sexual behaviour
- Pair-bonding
- Parental behaviour (paternal behaviour – lateral septum)
- Offensive aggression
- Social recognition (flank marking)
It has been suggested that a pluralistic neuroethological approach should greatly increase the power of future studies of Vasopressin and social behavior. Existing phenotypes are products of multiple interacting mechanisms. Goodson et al. (2001) develop a pluralistic neuroethological framework in order to ask the question: to what extent do the findings from one species yield accurate predictions for others? More relevantly, how do these findings relate to human physiology? Humans adapt differently to the seasons and there is no evidence to suggest that humans are in any sense seasonal breeders. If tropical Africa was the cradle of human evolution, as all the recent archaeological evidence suggests, there would have been no advantage for our early ancestors to restrict their births to certain season’s of the year; none of the great apes do.
Maybe it’s because man’s colonisation of the temperate and polar regions of the globe has been too recent, and our ability to control our environment so successful, that seasonality of reproduction has never had occasion to evolve. But given the right theoretical framework we can translate data across species and incorporate it into our knowledge of human physiology and behavior. We do indeed need to develop a different and perhaps new way of thinking, creating a broader framework in order to encompass all species.
The study of circadian rhythms is possibly one of the few disiplines of biology, neurochemistry and morphophysiology (as Professeur Paul Pevet has coined it) that attempts to study animals in context. The context of the circadian system is the day. Through adjustments in the light-dark cycle all characteristics of the phenotype have to be understood with respect to the other parts of the system. It is not sufficient to understand objects as individual entities or particulars which instantiate or ‘have’ properties. Researchers move beyond a narrow focus of searching for those attributes of the object that might help to explain and control its behaviour. This ontology, followed in many other areas of science, has meant that we have been able to discover many important scientific laws, but have failed to understand the fundamental nature of causality in the physical domain.
During my internship at the Laboratoire de Neurobiologie des Rythmes, I became particularly attracted to the pluralistic neuroethological approach of Bass and Goodson (2001) and was also influenced by the work of Edgar Morin. Then I read an interesting article in the Harvard University Gazette (September 25, 2003) where Marc Kirschner, (Carl W. Walter Professor of Cell Biology and the first chair of the Department of Systems Biology) said:
« As we understand more about the tiniest pieces that we are made of, it becomes increasingly clear that we do not understand how they work together as systems »
Things started coming together in my mind and I saw the potential of applying the pluralistic neuroethological approach to the study of humans. The approach recognises that the existing phenotypes of a species are products of multiple interacting mechanisms which include not only the structural and behavioural characters, but life history and ecological variables as well. It is a theoretical and methodological framework that extends the structure-function paradigm of much neurobiological research into a structure-function-context model more suitable for comparative studies and holistic research. Neuroanthropology, I came to understand, could be the Neuroethology of Homo sapiens sapiens.
« Science is totally dependent upon philosophical opinions for all of its goals and methods,
though it easily forgets this. »
Friedrich Nietzsche



Published by